HEALTH
With World Malaria Day set for tomorrow, scientists in Kampala are pulling back the curtain on how next-generation tools against malaria are built, tested and scrutinized years before they ever leave the lab.
The timing is urgent. The 2025 Africa Malaria Progress Report warns that momentum in the fight against malaria has stalled since 2015. Africa remains off track to meet the African Union’s 2030 elimination target, with only a handful of countries reaching key reduction milestones.
Experts point to a widening global funding gap, insecticide resistance, climate pressures, and fragile health systems as threats that could fuel a malaria resurgence without sustained investment and innovation.
Looking beyond nets and drugs
That challenge is pushing researchers to study complementary approaches alongside bed nets, insecticides, drugs, and vaccines. One of them: gene drive technology.
“Out of more than 3,500 mosquito species worldwide, only a small number transmit malaria,” researchers note. In sub-Saharan Africa, a few closely related species drive most transmission.
Scientists with Target Malaria are investigating whether gene drive technology could reduce populations of these malaria-carrying mosquitoes, or stop the parasite from passing from mosquito to human.
From embryo to colony — under the microscope
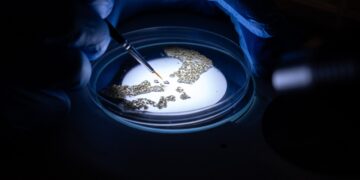
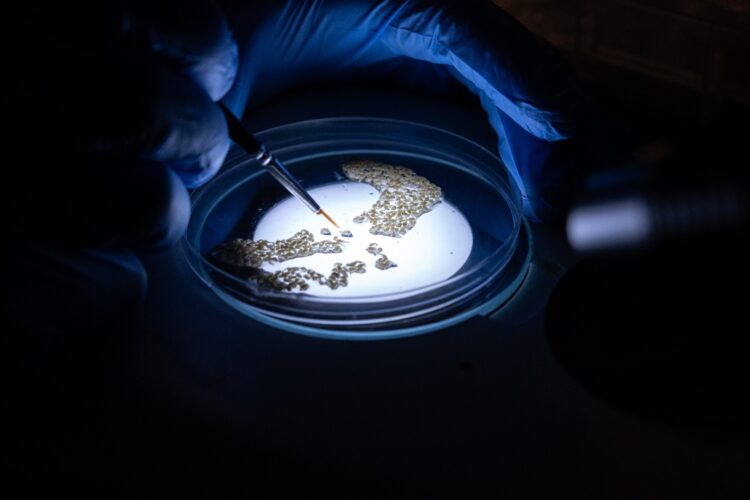
Developing gene drive mosquitoes is painstaking work. In highly controlled labs, researchers design genetic modifications and inject them into mosquito embryos with ultra-fine needles under microscopes. The window is tight: it must be done shortly after eggs are laid.
“Only few researchers in the world have this know-how,” the team says. The procedure is so delicate that many modified embryos fail. When it works, scientists identify the carriers and build lab colonies to track the trait across generations.
Testing, step by step
Once colonies are established, testing moves through stages. Small cage studies watch how gene drive mosquitoes interact and reproduce with wild-type mosquitoes of the same species. Larger indoor environments then mimic natural conditions to see how the modification might behave outside the lab.
Researchers compare lifespan, biting behaviour, disease transmission potential, and insecticide resistance against wild counterparts. Mathematical modelling runs alongside lab work to predict how the modification could spread and what impact it might have on malaria transmission.
“Innovation and investment are essential in the fight against malaria, but so is transparency,” says Dr Martin Lukindu, Post-Doctoral Research Associate with Target Malaria Uganda at the Uganda Virus Research Institute (UVRI). “People must understand how new technologies are researched, assessed and tested before they are ever considered for use.”
No mosquitoes in Africa yet
Dr Lukindu stresses that gene drive mosquitoes remain in the research phase. All current studies are in contained laboratories in Europe and the United States. “There are no gene drive mosquitoes in Africa.”
Any future use would require extensive safety studies, regulatory review in research countries, plus community engagement and approval from relevant authorities.
“As scientists, our goal remains the same: reduce malaria transmission and save African lives,” Dr Lukindu says.
For now, the message from Uganda’s labs is clear: with progress stalled and existing tools under pressure, new science is advancvie slowly, cautiously, and in public view.